John Dalton is one of the most well-known figures of the 18th Century. He was a physicist, chemist, and meteorologist. Throughout this career, Dalton worked on different disciplines such as color blindness, atomic theory, and even atomic laws. His theories and publications regarding atoms are still relevant today and played a huge role in shaping chemistry as seen today.
Although his career as compared to other 18th century scientists was a bit short-lived but it was sufficient enough for him to be named in the history books. So let’s discuss John Dalton and see what keeps his legacy alive even today.
Early Life and Career
John Dalton was born in Cumberland, England into a Quaker family. His father was a weaver and he received his education from both his father and a Quaker John Fletcher. Fletcher ran a small private school in a village nearby. Dalton would soon start earning for himself as his family at the time was too poor to support him. Therefore, at the age of 10, Dalton was offering his services to Elihu Robinson, who was a local wealthy Quaker.
At the age of 15, Dalton joined hands with his brother Jonathan to help him run a school in Kendal, Westmorland. At the time, it is thought that Dalton might have developed an interest in medicine or law. However, his relatives opposed his interest and was not able to attend English universities due to being a dissenter. As a result, Dalton acquired much of his scientific knowledge and information from John Gough who was a blind scientist but gifted in arts and sciences.
Then, at the age of 27, Dalton was hired as a mathematics teacher and natural philosophy at the “Manchester Academy”. He continued to offer his services for seven years until the academy underwent a serious financial crisis. This lead to his resignation and Dalton continued offering private tutorship in the same two subjects.
Meteorology
Dalton during his early days was greatly influenced by Elihu Robinson. At the time, Robinson was an instrument maker and a competent meteorologist who presented problems of meteorology and mathematics to Dalton, provoking his interest. While he was in Kendal, Dalton worked on and presented many solutions to problems in addition to answering questions in the Gentleman’s Diary and The Ladies Diary.
Then, at the age of 21 in 1787, Dalton began maintaining a meteorological diary in which he mentioned more than 200,000 observations. He rediscovered the theory of atmospheric circulation by George Hadley. In 1793, Dalton released his first publication Meteorological Observations and Essays. It contained several of his previous works. Although the publication was treated as original but the scholars of the time paid little to no attention to him.
Following the publication, Dalton’s second work, Elements of English Grammar was published in 1801.
Measuring Mountains
Upon his return from the Lake District, Dalton continued studying meteorology during holidays, much of which required hill-walking. At the time, there were no aeroplanes and weather balloons to study the temperature, humidity, and other factors related to weather. Therefore, the only way to do it was by climbing a mountain.
Dalton used a barometer to estimate the height. However, the maps for the Lake District were not published by the Ordnance Survey. With that being said, Dalton was one amongst few authorities who could climb the heights of the region’s mountains. Plus, he was often accompanied by Jonathan Otley who would study the heights of the local peaks as well and then compare his figures with Dalton’s. Later, Otley turned into a close friend and assistant of Dalton.
Color Blindness
Dalton shortly after arriving in Manchester in 1794, was elected as a member of the Manchester Literary and Philosophical Society. A few days later he published his first paper on “Extraordinary facts relating to the vision of colors”. He stated that the color shortage perception is due to the eyeball’s liquid medium discoloration. Since both Dalton and his brother were colorblind, he stated that the condition might be hereditary.
Even though Dalton’s theory did not receive much attention while he was alive but after he passed away, his broad research and methodical nature received so much attention that Daltonism was used as a common term for color blindness. In 1995, a thorough study of Dalton’s preserved eyeball suggested that he might have had a less common type of color blindness. As a result, he was only able to recognize the yellow color.
Atomic Theory
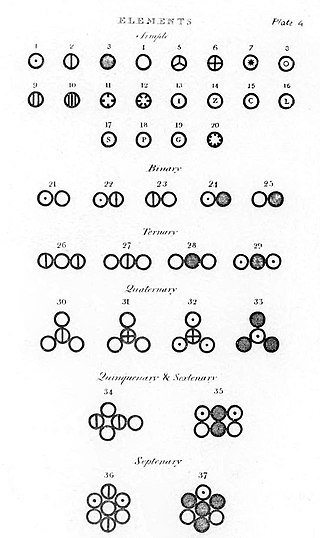
Dalton’s most famous and important work is related to atomic theory in chemistry. Although it is known that Dalton spent most of his life studying atoms but the origin of Dalton’s atomic theory is not known. It might be that his theory originated from researches on ethylene, methane, or by analysis of nitrogen dioxide and nitrous oxide.
From 1814 to 1819, William Higgins, an Irish Chemist had stated that Dalton had plagiarized his idea but Higgin’s work did not contain any information related to atomic mass. However, recent claims suggest that Dalton’s development of thought might have been triggered by Bryan Higgins, who was another Irish Chemist and uncle of William Higgins.
Bryan believed that an atom is a heavy central particle surrounded by a caloric atmosphere, which was assumed a heat substance at the time. With that being said, it was the diameter of the caloric atmosphere that decided the size of the atom. Therefore, based on the evidence, it is clear that Dalton was well-aware and familiar with Bryan’s theory but he never acknowledged Bryan’s caloric model anticipation as well.
On the other hand, Dalton’s fundamental role in atomic theory was that he determined and provided a method calculating atomic weights for the chemical elements. It was something neither William nor Bryan Higgins did. In addition to that, Dalton’s laboratory notebooks discovered in the rooms of the Manchester Literary and Philosophical Society prove that Dalton’s study and concept of atoms arose when he began studying the atmosphere’s physical properties and other gases.
Atomic Weights
Dalton released his first table of relative atomic weights featuring six elements, relative to a hydrogen atom’s weight, conventionally taken as 1. Since these are relative weights, there are no units of weights attached to them. Although Dalton never mentioned how he came up with these numbers but according to his laboratory notebook dated 6 September 1803, he set the relative weights of the atoms of a number of elements after analyzing ammonia, water, and carbon dioxide, etc.
Dalton’s law of multiple proportions was a direct result of the extension of his idea to general substances. His experiments offered excellent results by confirming his deduction.
Experimental Approach
Dalton was an individual who was often satisfied with inaccurate and rough estimates even though better ones were attainable. According to Sir Humphry Davy, Dalton was a “very coarse experimenter” who would always come up with the results he required and trusted his head more than the hands.
In addition to that, historians who have conducted experiments the same way Dalton did, swear by his precision and skill. Dalton had mentioned in one of his books that he would often take others results for granted and was misled. However, he also added that he trusted his personal experience more than what others had to offer. At the same time, his decision to independently carry out researches and publications did not receive any attention or value.
Moreover, Dalton carried unconventional views regarding Chlorine, even though Davy had settled its elementary character. Dalton continued using the atomic weights even when other techniques and more accurate determinations by other chemists had superseded his concept.
Public Life
Dalton even before introducing his atomic theory had gained quite a bit of reputation. In 1803, he was asked to offer lectures on natural philosophy at the Royal Institution in London, while delivering additional lectures in 1809-1810. Some witnesses stated that as a lecturer, he lacked several qualities. In other words, he was harsh indistinct in voice.
Then, in 1810, Sir Humphry Davy asked Dalton to offer himself as a candidate for the fellowship of the Royal Society but he declined perhaps due to financial reasons. As a result, in 1822 Dalton without his knowledge was proposed and paid the usual feel on the election. Apart from that, Dalton had been a member of the French Academie des Sciences for six years. In 1834, he was also elected as a Foreign Honorary Member of the American Academy of Arts and Sciences.
Final Word
Dalton during his last days continued performing experiments even though he had suffered a minor stroke. He passed away in 1844 while the state carried out a civic funeral with full honors. His legacy was such that more than 40,000 people filed past his coffin. He had also instructed his eyes to be examined after his death. This helped the researchers study more about the man who followed his interest and love for experiments until his last breath.